|
So let me go ahead and draw in deuterium's one neutron. Deuterium is hydrogen, so it must have one proton in the nucleus and it must have one electron outside the nucleus, but if you look at the definition for isotopes, atoms of a single element that differ in the number of neutrons, protium has zero neutrons in the nucleus. Let me go ahead and write deuterium here. So let's talk about the next isotope of hydrogen. That differ in the number of neutrons in their nuclei. An isotope, isotopes are atoms of a single element. So this is protium and let's talk about isotopes. So this is one, this one version of hydrogen. 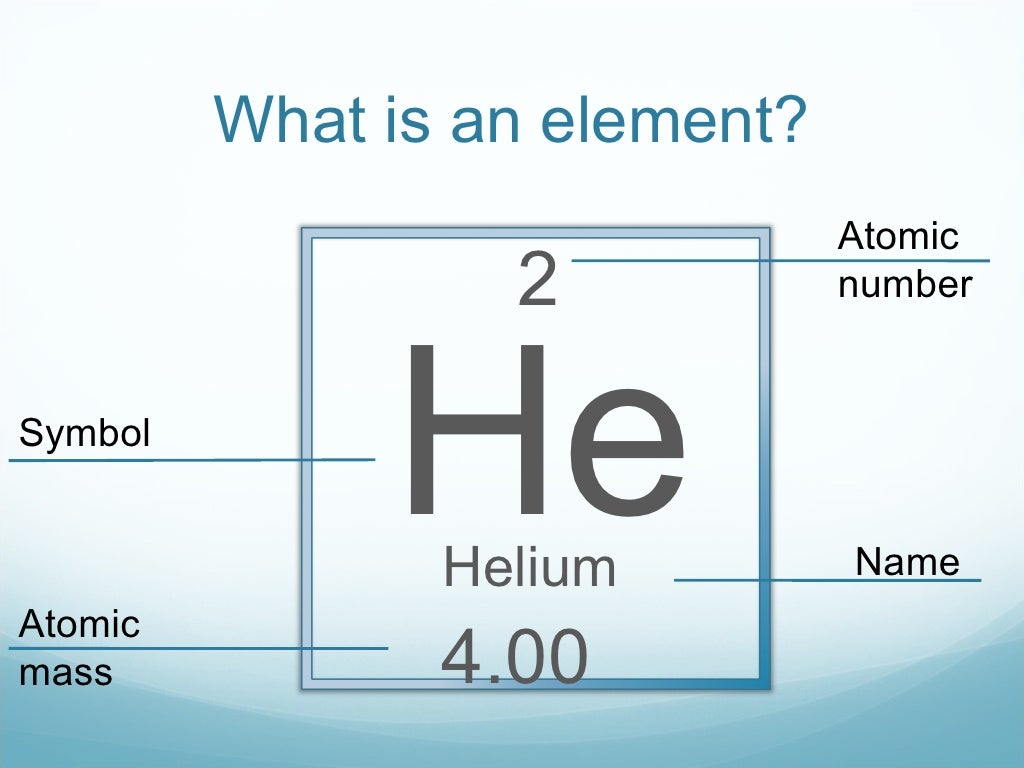
So this isn't actually what an atom looks like, but it's a very simply view that helps you get started. 
I'm going to draw that one electron somewhere outside the nucleus and I'm going to use the oversimplified Bohr model. So there's my one proton in the nucleus, and we're talking about a neutral hydrogen atom, so there's one electron. We know the atomic number of hydrogen is one, so there's one proton in the nucleus. So let's go ahead and draw an atom of hydrogen. In a neutral atom, the number of protons is equal to the number of electrons, because in a neutral atom there's no overall charge and the positive charges of the protons completely balance with the negative charges of the electrons. So it's right here, so there's one proton in the nucleus of a hydrogen atom. So for hydrogen, hydrogen's atomic number is one. So we're going to talk about hydrogen in this video. 
And you can find the atomic number on the periodic table. So the atomic number is symbolized by Z and it refers to the number of protons in a nucleus.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
